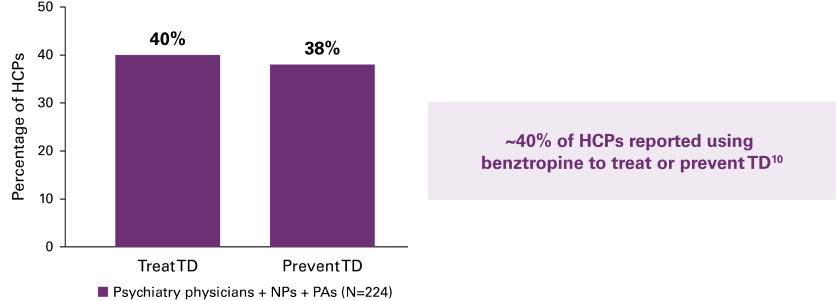Tardive dyskinesia (TD) is a persistent, typically irreversible, hyperkinetic movement disorder, resulting from chronic exposure to antipsychotics that affects approximately 785,000 patients in the United States.1-4
The term "extrapyramidal symptoms" (EPS) has a long history of being used to describe all drug-induced movement disorders (DIMDs) experienced by patients taking antipsychotics.1,5,6 These include acute syndromes, developing within hours or days of treatment (eg, dystonia, akathisia, and drug-induced parkinsonism [DIP]) as well as tardive syndromes, occurring after a continued period of treatment (eg, TD, tardive dystonia, and tardive akathisia).7 Although these syndromes are often lumped together, they are each distinct movement disorders, with different underlying causes and, consequently, different treatment approaches. For instance, TD and DIP are common movement disorders occurring in up to 1 of 3 patients taking antipsychotics, but their distinction may often be misunderstood.1,8,9 However, treatment for DIP can worsen TD and vice versa.1,9 Therefore, it is critical to differentiate TD from other DIMDs, particularly DIP, in order to guide appropriate treatment for patients.
Psychiatric clinicians continue to use anticholinergics broadly to treat or prevent EPS.10 This can negatively impact patients with TD. While anticholinergics are appropriate for treating DIP and acute dystonia, they can make TD movements worse.1,9 In fact, the prescribing information for benztropine, a widely prescribed anticholinergic, warns that antiparkinsonism agents do not alleviate symptoms of TD and may, in some instances, exacerbate them.11
As a result, the American Psychiatric Association (APA) treatment guidelines and the Diagnostic and Statistical Manual of Mental Disorders, 5th ed, Text Revision (DSM-5-TR) warn that anticholinergics should not be used to prevent or treat TD.12,13 Instead, vesicular monoamine transporter 2 (VMAT2) inhibitors are recommended as first-line treatment for TD that has an impact on the patient, regardless of the severity of movements.13
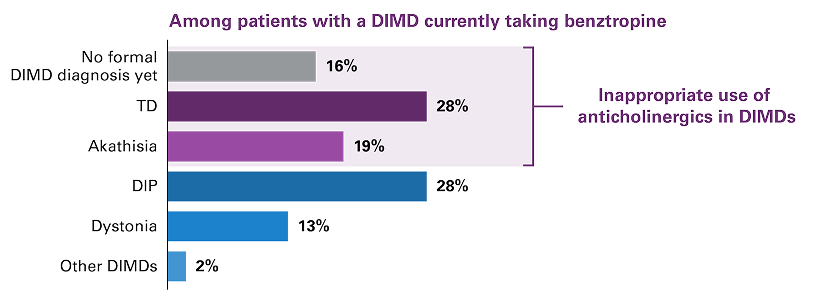
Despite these recommendations, a significant number of patients with TD continue to be treated with anticholinergic medications, reflecting a critical need for increasing awareness of this issue.10 Results from a survey of high prescribers of benztropine for TD (33 psychiatrists and 13 nurse practitioners [NPs]/physician assistants [PAs]) found that 30% of patients with a DIMD were currently taking benztropine.4 Among these patients, up to 63% were inappropriately prescribed anticholinergics, including 28% of patients with TD (Figure 1).4
Additionally, this report demonstrated that healthcare professionals (HCPs) most frequently use benztropine long-term, with up to 93% of patients with TD being on benztropine for more than 3 months.4 While ~50% of survey respondents reported that they were very familiar with tardive syndromes or agreed with statements related to differentiated diagnosis of TD and DIP, and 37% understand that anticholinergic drugs are not effective for the treatment of TD, less than one-third seemed concerned that prescribing them may exacerbate TD and would discontinue anticholinergic treatment if they suspected a patient had TD.4

In a second survey by Chepke et al, ~40% of psychiatry HCPs (151 psychiatrists and 98 NPs/PAs) reported using benztropine to prevent or treat TD (Figure 2).10 Additionally, they were more inclined to continue using benztropine for more than 6 months, with a significantly higher percentage continuing for more than 12 months.10
These studies demonstrate that even with a proper diagnosis of TD, patients are still frequently prescribed benztropine, highlighting that broad and inappropriate use of anticholinergics in patients with TD persists in psychiatry, despite recommendations that it can worsen symptoms.
Compiled results from the behaviors and attitudes of high prescribers of benztropine for TD indicated that patients reporting it as helpful, and multiple or unconfirmed diagnoses were among the most frequent reasons for prescribing anticholinergics (Figure 3).4
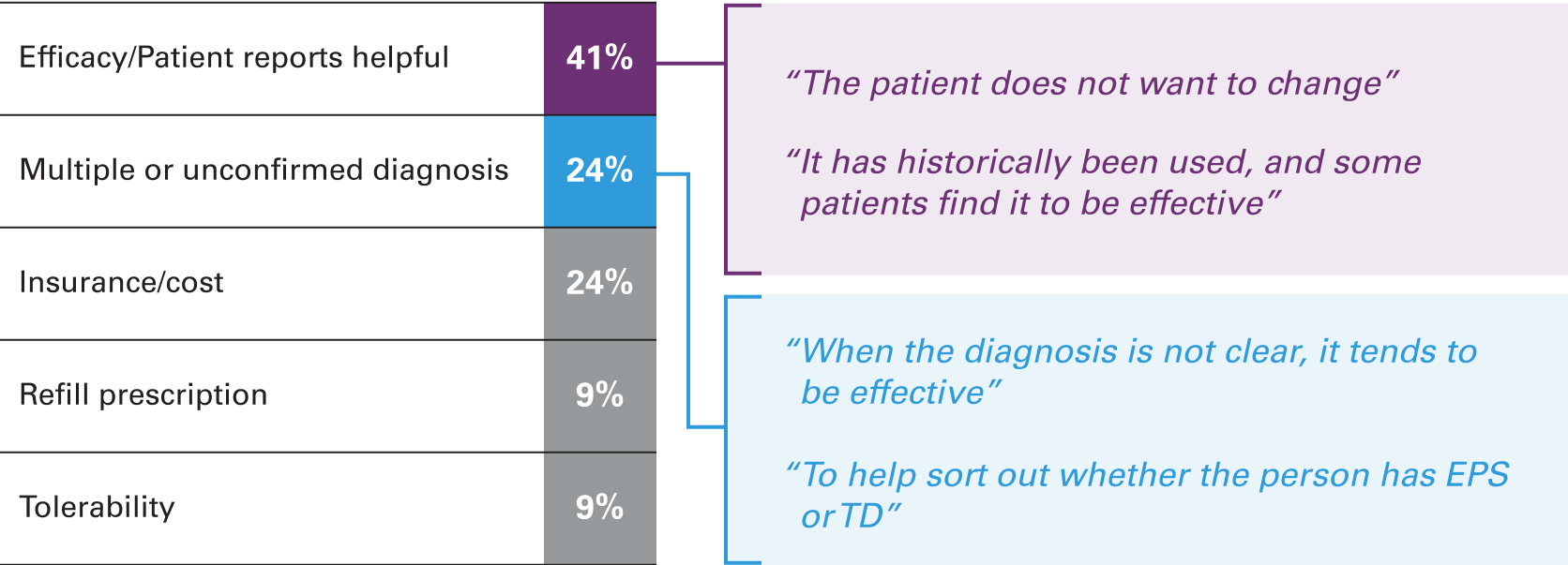
Another possible reason outlined in this report was that respondents were following teaching habits learned during medical school or from more experienced attending physicians who endorse the use of anticholinergics for any movement disorder.4 Additionally, some prescribers may be unwilling to deprescribe the medication, fearing it would hurt their relationship with patients.4 Others lack motivation to change prescribing habits.4 Finally, providers may still prescribe anticholinergics over standard-of-care therapies due to the lack of understanding of APA recommendations around the treatment of TD. In fact, Chepke et al found that less than 40% of psychiatric HCPs were somewhat or very familiar with the APA 2020 guidelines.10 However, of HCPs who reported being at least somewhat familiar, the majority agreed that VMAT2 inhibitors are the first-line treatment for TD.10
Educating clinicians on the importance of deprescribing anticholinergics in patients with TD is required and has previously been accomplished in a community mental health setting.14 Critical reasons to consider deprescribing anticholinergics include that they are not indicated to treat or prevent TD, can make TD symptoms worse, and are inappropriate for many older adults.11-13,15 For patients taking an anticholinergic, it is recommended to gradually reduce anticholinergic therapy to lessen the potential impact on TD symptoms.9 VMAT2 inhibitors are the only FDA-approved treatment for TD and are recommended as first-line treatment for TD that has an impact on the patient, regardless of severity of movements.12
Although the APA treatment guideline and DSM-5-TR caution against using anticholinergics, such as benztropine, to treat or prevent TD, anticholinergics are still being overused in psychiatry settings, because of patients reporting their use as helpful, inertia (eg, absence of motivation to change prescribing habits), lack of understanding of APA recommendations, or self-perpetuating prescribing patterns from more senior staff and attending physicians who support the use of anticholinergics for any movement disorders4,10,12,13
The APA recommends that patients who have TD that has an impact on the patient be treated with a reversible inhibitor of the VMAT2, regardless of the severity of movements13
Implementing standard protocol or guidelines in clinical practices may influence prescribing patterns toward deprescribing anticholinergics and implementing habits for the use of VMAT2 inhibitors for patients with TD