Tardive dyskinesia (TD) and drug-induced parkinsonism (DIP) are distinct movement disorders. Both are caused by exposure to antipsychotics but they have different underlying causes and different treatment approaches.1,2 Vesicular monoamine transporter 2 (VMAT2) inhibitors are recommended by the American Psychological Association (APA) as a first-line treatment for TD that has an impact on the patient regardless of severity of movements, and are the only treatment for TD approved by the US Food and Drug Administration (FDA).3 In contrast, anticholinergics that are used to treat DIP are not approved for TD.4,5 Despite this, a significant number of patients with TD are treated inappropriately with anticholinergics.6
Because treatment for one condition can worsen the other, it is critical to differentiate TD from DIP and treat each appropriately.1,2
The efficacy and safety of AUSTEDO in the treatment of TD was evaluated in 2 pivotal clinical trials, ARM-TD and AIM-TD.7,8
ARM-TD was a randomized, double-blind, parallel-group study in adult patients with TD. In this flexible-dose clinical trial, patients’ doses were titrated to an individualized dosage that reduced abnormal movements and was tolerated by the patient. Patients were randomized 1:1 to receive AUSTEDO twice daily (BID) or placebo.7
AIM-TD was a randomized, double-blind, placebo-controlled, phase 3 clinical trial in adult patients with TD. Patients were randomly assigned 1:1:1:1 to receive 1 of 3 fixed doses of AUSTEDO (12 mg/day, 24 mg/day, or 36 mg/day) or placebo.8
For both ARM-TD and AIM-TD, the primary efficacy endpoint was change in Abnormal Involuntary Movement Scale (AIMS) score from baseline to Week 12.7,8
RIM-TD was an open-label study evaluating the efficacy of AUSTEDO as a long-term maintenance therapy in patients with TD who successfully completed either the ARM-TD or AIM-TD clinical trials. Enrolled patients discontinued the study drug for at least 1 week before beginning AUSTEDO (12 mg/day) and titrated for up to 6 weeks. The dose was adjusted once per week to identify a dose that adequately controlled dyskinesia and was tolerated by the patient.9
Patients receiving AUSTEDO in both the ARM-TD and AIM-TD studies achieved significant improvement in AIMS total scores compared with patients receiving placebo.7,8 Data did not suggest substantial differences in efficacy across various demographic groups.4
In the ARM-TD study, patients receiving AUSTEDO achieved a reduction of 3.0 points from baseline to Week 12 compared with 1.6 points for patients receiving placebo, and a treatment effect of -1.4 points (P=0.019) when compared with placebo (Figure 1). The mean dose of AUSTEDO at the end of the treatment period was 38.3 mg/day.7
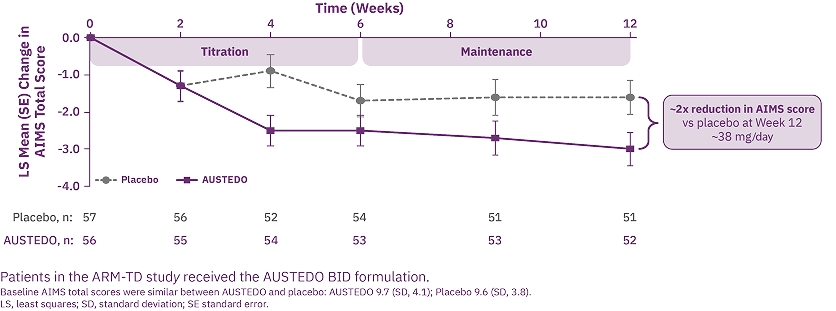
The results from ARM-TD were consistent with the results in AIM-TD at Week 12. In AIM-TD, patients receiving AUSTEDO 36 mg/day achieved a reduction of 3.3 points from baseline to Week 12, compared with 1.4 points for patients receiving placebo, and a treatment effect of -1.9 points (P=0.001) when compared with placebo.8
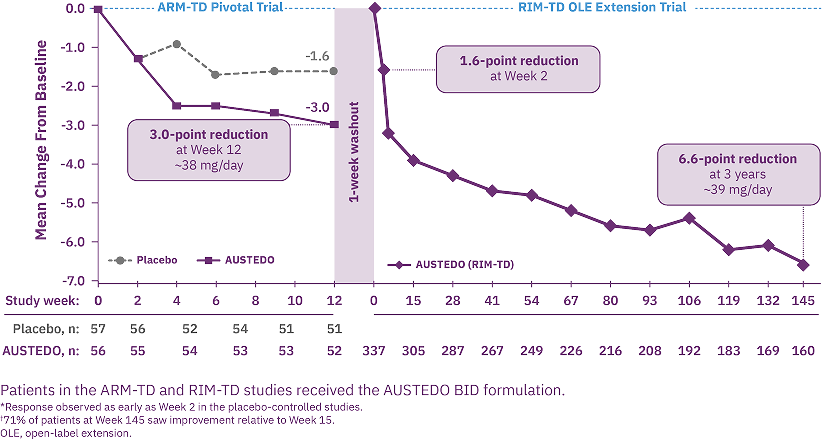
During the overall treatment period, a reduction in mean AIMS total scores was observed from baseline through Week 145, with 67% of patients achieving ≥50% improvement in AIMS score through year 3 (Figure 2).9
The most common adverse reactions occurring in patients treated with AUSTEDO (>3% and greater than placebo) were nasopharyngitis and insomnia. One or more adverse reactions resulted in a reduction of the dose of study medication in 4% of patients treated with AUSTEDO and in 2% of patients treated with placebo.4 The adverse reactions occurring in patients treated with AUSTEDO (12-48 mg per day) (≥2%) are summarized in Figure 3.
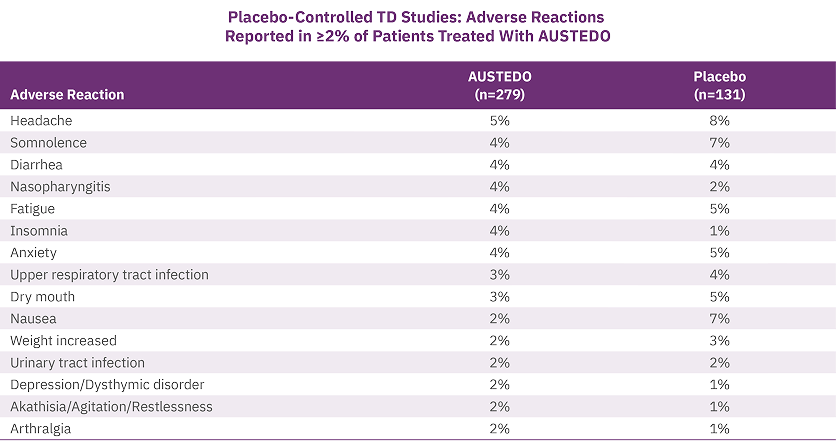
Once patients were titrated to their maintenance dose, the following adverse reactions were no longer reported: dry mouth and nausea in AIM-TD and somnolence and dry mouth in ARM-TD.10 Adverse reactions in the 3-year RIM-TD study were comparable to the 12-week ARM-TD and AIM-TD studies, with no new safety signals identified over the course of the study.9
Adverse reactions with AUSTEDO XR are expected to be similar to those with AUSTEDO BID.4
To limit possible drug-drug interactions, it is important to consider metabolic pathways when choosing a treatment for TD, particularly in patients who are taking medications for comorbidities. AUSTEDO XR is metabolized primarily by CYP2D6, with minor contributions from CYP3A4/5 and other enzymes to form several minor metabolites.4 It has a range of dosing options for patients on concomitant medications and has the most dosing options for patients taking common strong cytochrome P450 (CYP) inhibitors or inducers (Figure 4).4,11 Even patients taking strong CYP inducers or inhibitors can titrate up to 36 mg/day, and no dose adjustments to P-gp substrates (eg, calcium channel blockers, statins, and antimicrobials) are required when taking AUSTEDO XR.4,12

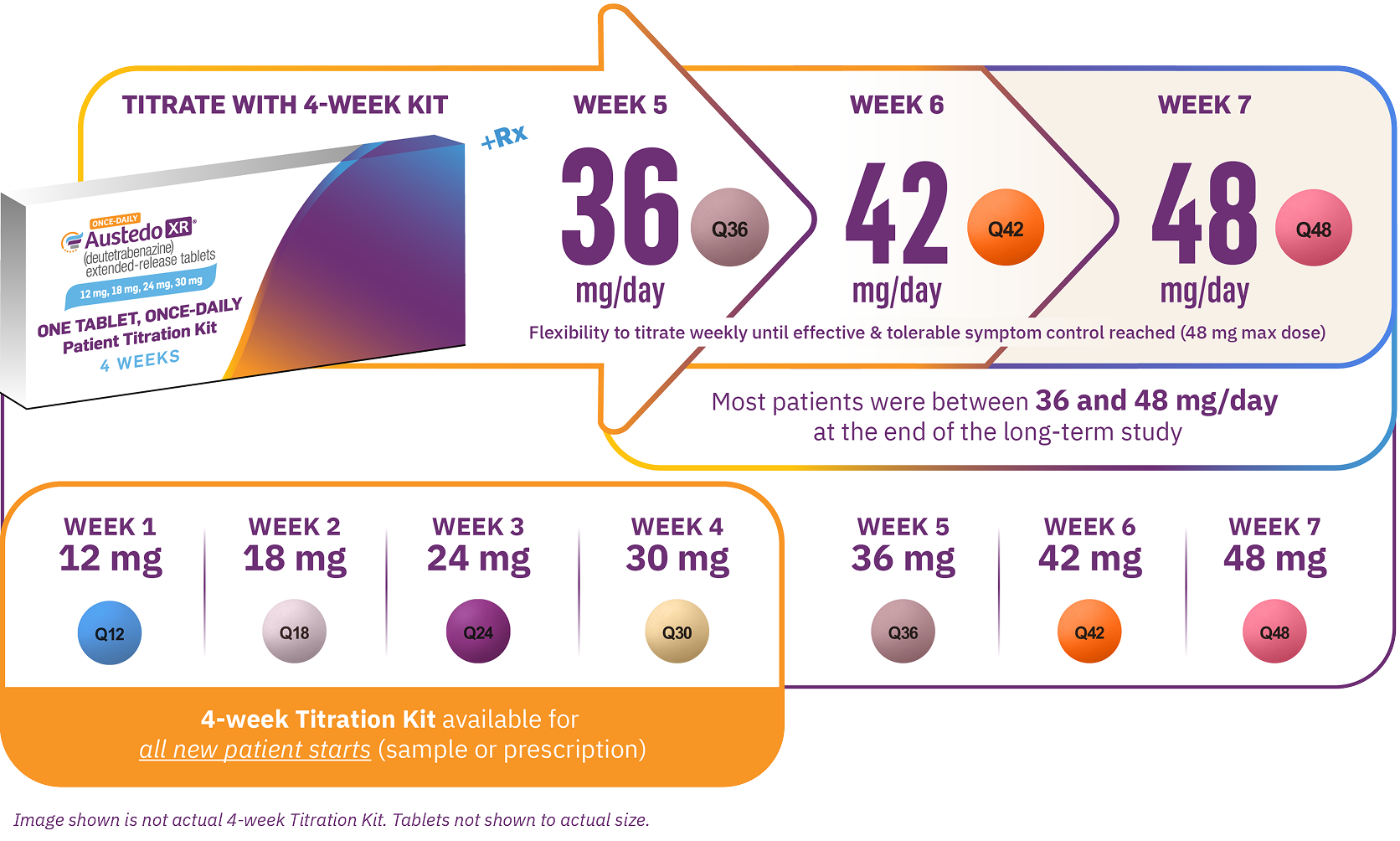
AUSTEDO XR is available as a once-daily, extended-release tablet. The recommended starting dose of AUSTEDO XR for patients with TD is 12 mg/day and may be increased at weekly intervals by 6 mg/day, providing the flexibility to titrate as needed to achieve effective and tolerable symptom control. The maximum recommended daily dosage is 48 mg (Figure 5).4
In a real-world survey, 98% of patients treated with once-daily AUSTEDO XR reported that taking it was very easy, and 95% of patients reported that they planned to continue treatment with AUSTEDO XR.13
In addition to patients reporting satisfaction with using AUSTEDO XR, patients also reported improvements in other areas of life as a result of movement reduction, including greater self-esteem, less embarrassment and anxiety, and overall better emotional well-being. Patients also reported feeling more comfortable with social activities, including spending time with family and friends, speaking to others, and going to work and school.13
VMAT2 inhibitors are the recommended first-line treatment for patients with TD.3 It is critical to differentiate TD from other movement disorders and treat it appropriately.4,5 It is also important to consider the metabolic pathways for all of a patient’s prescribed treatments when identifying an appropriate treatment.1,2 AUSTEDO XR is a VMAT2 inhibitor that has demonstrated efficacy and safety in the treatment of TD, with no recommendations against concomitant use with CYP3A4/5 inducers or inhibitors.4,7,8 The flexible, once-daily dosing possible with AUSTEDO XR allows for individualized treatment to achieve effective and tolerable control.4
1. Ward KM, Citrome L. Antipsychotic-related movement disorders: drug-induced parkinsonism vs. tardive dyskinesia—key differences in pathophysiology and clinical management. Neurol Ther. 2018;7(2):233-248. 2. Hauser RA, Meyer JM, Factor SA, et al. Differentiating tardive dyskinesia: a video-based review of antipsychotic-induced movement disorders in clinical practice. CNS Spectr. 2022;27(2):208-217. 3. American Psychiatric Association. The American Psychiatric Association Practice Guideline for the Treatment of Patients With Schizophrenia. 3rd ed. Washington, DC: American Psychiatric Association; 2021. 4. AUSTEDO XR® (deutetrabenazine) extended-release tablets/AUSTEDO® current Prescribing Information. Parsippany, NJ: Teva Neuroscience, Inc. 5. Benztropine mesylate tablets. Prescribing Information. Warren, NJ: Cipla USA, Inc. 6. Chepke C, Benning B, Cicero S, et al. Investigating real-world benztropine usage patterns in movement disorders: claims analysis and health care provider survey results. Prim Care Companion CNS Disord. 2023;25(4):22m03472. 7. Fernandez HH, Factor SA, Hauser RA, et al. Randomized controlled trial of deutetrabenazine for tardive dyskinesia: the ARM-TD study. Neurology. 2017;88(21):2003-2010. 8. Anderson KE, Stamler D, Davis MD, et al. Deutetrabenazine for treatment of involuntary movements in patients with tardive dyskinesia (AIM-TD): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Psychiatry. 2017;4(8):595-604. 9. Hauser RA, Barkay H, Fernandez HH, et al. Long-term deutetrabenazine treatment for tardive dyskinesia is associated with sustained benefits and safety: a 3-year, open-label extension study. Front Neurol. 2022;13:773999. 10. Data on file. Teva Neuroscience, Inc. Parsippany, NJ. 11. Ingrezza® (valbenazine) capsules. Prescribing Information. San Diego, CA: Neurocrine Biosciences, Inc. 12. Drug Bank Online: P-glycoprotein substrates. https://go.drugbank.com/categories/DBCAT002668 13. Jain R et al. Real-world evidence of patient experience with once-daily deutetrabenazine extended-release tablets for tardive dyskinesia and chorea in Huntington disease in the United States. Presented at: Psych Congress; October 29-November 2, 2024; Boston, MA.








