Tardive dyskinesia (TD) and drug-induced parkinsonism (DIP) are movement disorders resulting from exposure to dopamine receptor blocking agents, such as antipsychotic medications. However, each disorder has distinct underlying pathophysiology and specific clinical presentation and requires different treatment approaches.1,2 While anticholinergic medications can be used to treat the symptoms of DIP, they are not effective in treating TD and may even worsen hyperkinetic symptoms.1 Vesicular monoamine transporter 2 (VMAT2) inhibitors, such as AUSTEDO XR, are the only US Food and Drug Administration (FDA)-approved treatment for adult patients with TD.3
This article reviews key efficacy and safety data from the pivotal clinical trials of AUSTEDO and dives into the range of available dosing options, including recommendations for specific populations and patients taking concomitant medications. Also included are data from real-world surveys of patient-reported experiences on AUSTEDO XR.
The precise mechanism by which deutetrabenazine exerts its activity is unknown but is believed to be related to its effect as a reversible depletor of monoamines, including dopamine, from nerve terminals (Video).4
The efficacy and safety of AUSTEDO in the treatment of TD was evaluated in 2 pivotal clinical trials, ARM-TD (Aim to Reduce Movements in Tardive Dyskinesia) and AIM-TD (Addressing Involuntary Movements in Tardive Dyskinesia)5,6:
- ARM-TD – Flexible-dose study (N=113): 12-week, randomized, double-blind, placebo-controlled phase 2/3 trial in which doses were titrated to an individualized dose that reduced abnormal movements and was tolerated. After screening, patients were randomized 1:1 to receive AUSTEDO twice daily (BID) or placebo6
- AIM-TD – Fixed-dose study (N=222): 12-week, randomized, double-blind, placebo-controlled phase 3 trial in which patients were randomized to receive placebo or AUSTEDO (12 mg, 24 mg, or 36 mg)4,5
The primary efficacy endpoint in both trials was the change in Abnormal Involuntary Movement Scale (AIMS) total score from baseline to Week 12.4-7
AUSTEDO was not studied in patients currently taking or who had recently (within 30 days) received anticholinergic medications. However, patients with prior anticholinergic use were not excluded from the study.7 Excluding patients currently taking anticholinergics aligns with TD treatment guidelines and warnings in the label of benztropine, a common anticholinergic, which states that it does not alleviate TD symptoms and may aggravate them.3,8
In these placebo-controlled trials, patients who received AUSTEDO demonstrated a rapid response as early as Week 2 and achieved a significant and meaningful reduction in TD severity at Week 12.4,6,7
- In ARM-TD, patients receiving AUSTEDO had an AIMS score reduction of 3.0 points from baseline to Week 12, compared with 1.6 points in the placebo group (treatment effect of -1.4 points, P=0.019) (Figure 1, left graph).6,7 At Week 12, 94% of patients were taking a dose of ≥30 mg/day, and the average daily dose was ~38 mg6,7
- In AIM-TD, AUSTEDO led to an AIMS total score reduction of 3.3 points from baseline in the 36 mg/day arm compared with a reduction of 1.4 points with placebo at Week 12 (treatment effect of -1.9 points, P=0.001)4,5
Patients on a stable dose of an antipsychotic maintained that medication while on AUSTEDO.5,6
RIM-TD (Reducing Involuntary Movements in Participants With TD), a single-arm, open-label extension (OLE), long-term maintenance study, was designed to evaluate long-term treatment with AUSTEDO for up to 3 years.9 Following completion of ARM-TD or AIM-TD, patients were eligible to enter the RIM-TD study.9 Among the patients evaluated, 337 patients were in treatment at baseline and 160 patients remained on treatment through the end of Week 145.9 The mean overall compliance rate, assessed by pill counts, was nearly 90% at 3 years.7
Increased improvement in AIMS total score was observed over 3 years in the longest TD clinical trial to date (OLE), with 71% of patients at Week 145 seeing improvement relative to Week 15 (Figure 1, right graph). At Week 145, 87% of patients were taking a dose of ≥30 mg/day and the average daily dose was ~39 mg.7,9
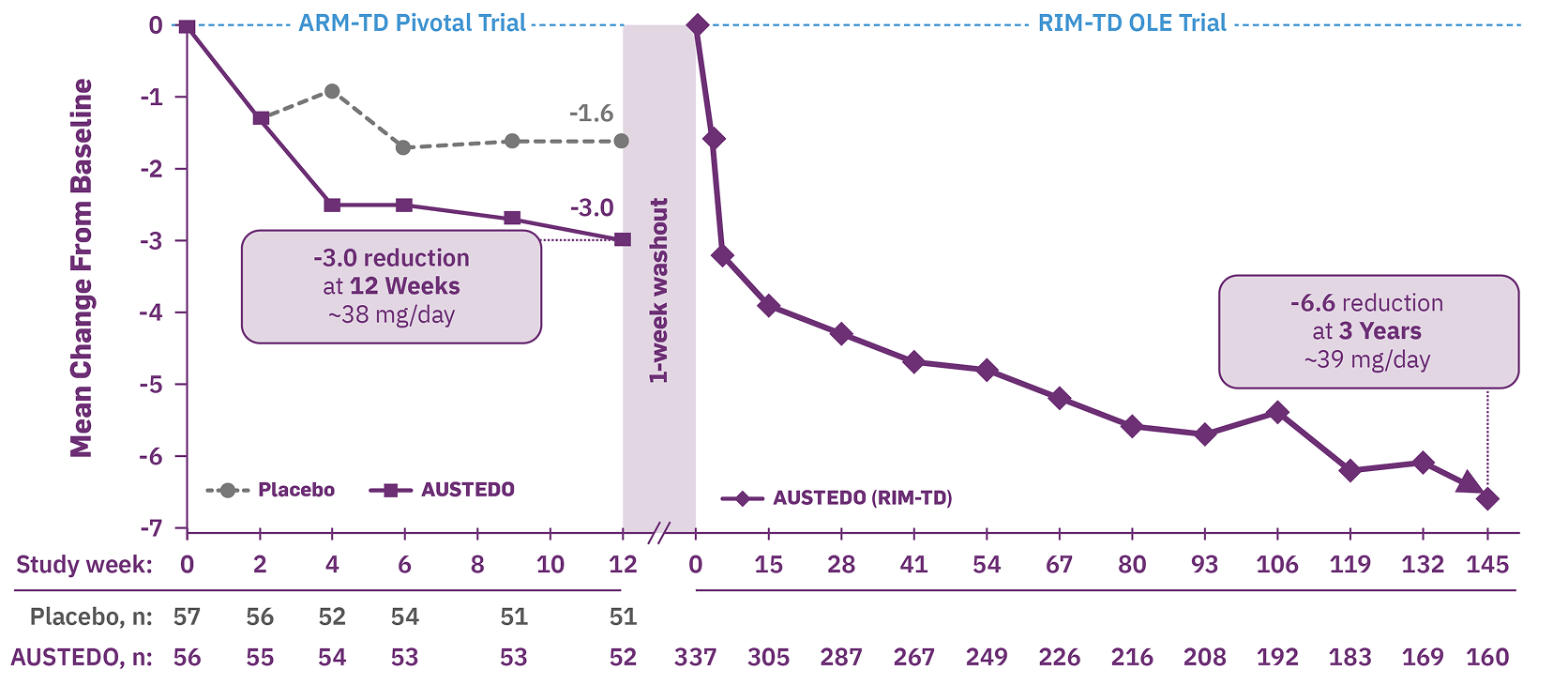
Baseline AIMS score: ARM-TD (placebo: 9.6 [SD, 3.8]; AUSTEDO: 9.6 [SD, 4.1]) and RIM-TD (AUSTEDO: 10.7 [SD, 4.7]).
ARM-TD, Aim to Reduce Movements in Tardive Dyskinesia; OLE, open-label extension; RIM-TD, Reducing Involuntary Movements in Participants With TD.
AUSTEDO is the only VMAT2 inhibitor demonstrating 3-year results in patients at increased risk of TD, including in (Figure 2)10-12
- Patients ≥65 years
- Postmenopausal women
- Patients with mood disorders and schizophrenia

Patients in the ARM-TD and RIM-TD studies received the AUSTEDO BID formulation.
*Includes patients with bipolar disorder and depression.
†Includes schizophrenia or schizoaffective disorder.
AIMS, Abnormal Involuntary Movements Scale.
The most common adverse reactions reported across both placebo-controlled studies in patients with TD treated with AUSTEDO (>3% and greater than placebo) were nasopharyngitis and insomnia.4 Adverse reactions occurring in patients treated with AUSTEDO (12-48 mg/day) (≥2%) are summarized in Figure 3.
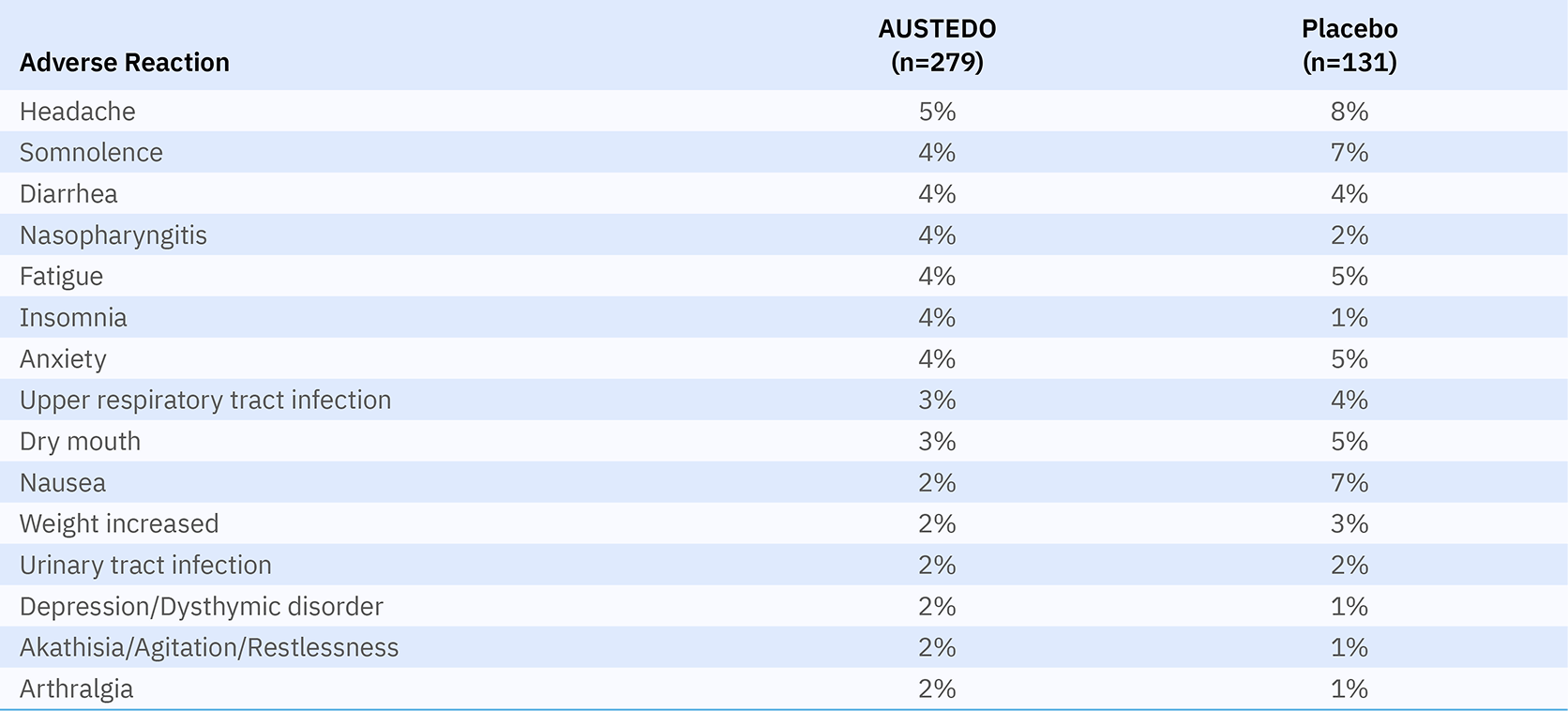
Discontinuation due to adverse events (AEs) occurred in 4% of patients taking AUSTEDO compared with 3% of patients treated with placebo.5 Four percent of patients required dose reduction of AUSTEDO due to AEs compared with 2% of patients taking placebo.4 Once patients were titrated to their maintenance dose, several AEs were no longer reported, including dry mouth and nausea in AIM-TD and somnolence and dry mouth in ARM-TD.7
No new safety signals were identified in RIM-TD, and AEs were comparable with those in the 12-week clinical trials.9
It is important to highlight that the safety and tolerability of AUSTEDO was consistent in older and younger patients, in postmenopausal women, and in patients with underlying psychotic or mood disorders.10-12
Adverse reactions with AUSTEDO XR are expected to be similar to AUSTEDO BID.4
AUSTEDO XR is available in 12 mg, 18 mg, 24 mg, 30 mg, 36 mg, 42 mg, and 48 mg extended-release tablets, providing dosing flexibility for effective and tolerable symptom control.4 The recommended starting dose of AUSTEDO XR for patients with TD is 12 mg/day, which may be increased at weekly intervals by 6 mg/day.4 The average daily dose in clinical trials was >36 mg/day, and 52% of patients were taking 36 mg/day, 42 mg/day, or 48 mg/day at Week 145 in the long-term study.5,9 Additionally, in pharmacokinetic studies, increased plasma levels of AUSTEDO correlated with higher potential for treatment success, but not AEs.13,14
AUSTEDO XR should be swallowed whole. Tablets should not be chewed, crushed, or broken. AUSTEDO XR may be taken with or without food (Figure 4).4 If a patient misses a dose for less than 1 week, they can restart at the same dose of AUSTEDO XR or AUSTEDO.4
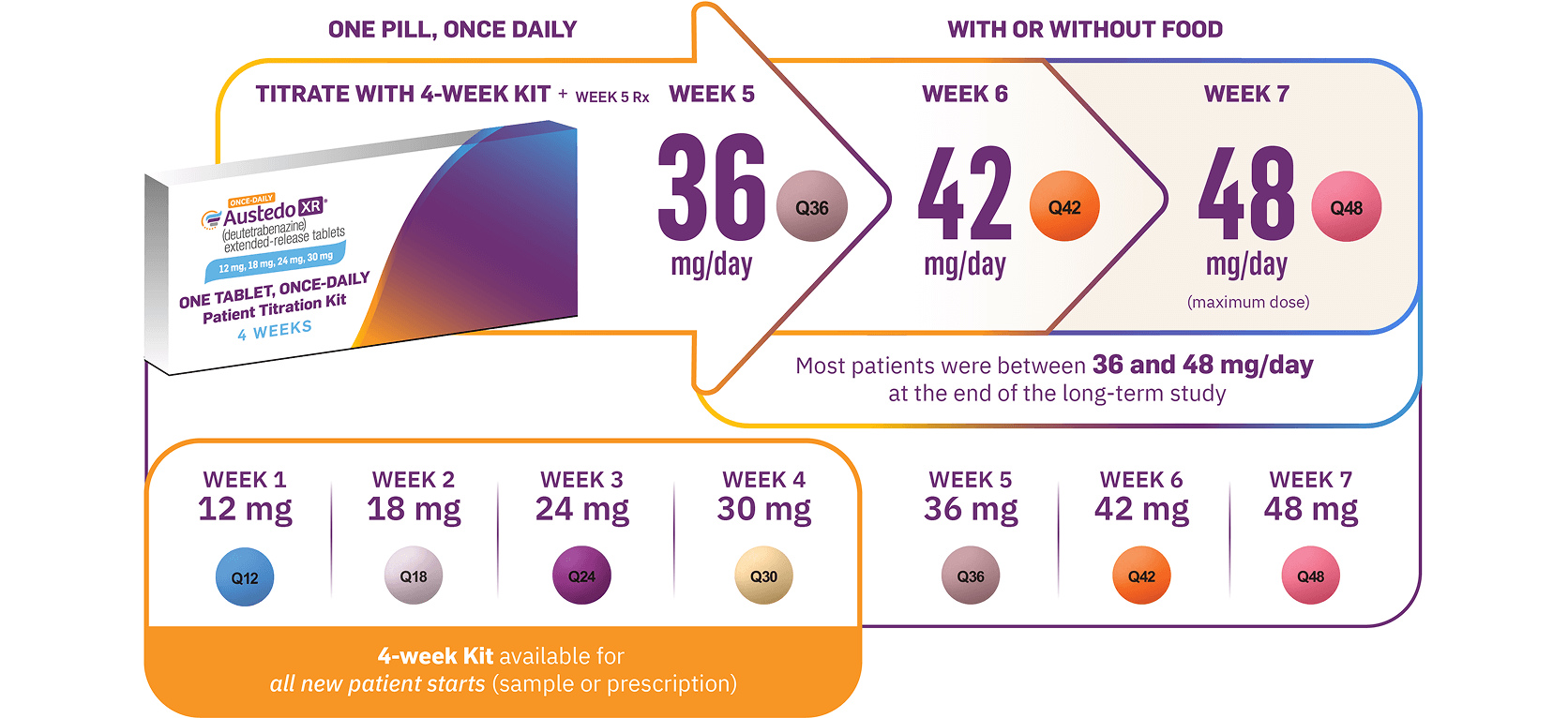
Image shown is not the actual 4-week Titration Kit. Tablets not shown to actual size.
Patients with TD often take medications for both nonpsychiatric comorbidities and mental health stabilization. Therefore, it is critical for clinicians to be aware of these regimens and consider metabolic pathways to reduce the risk of drug-drug interactions (DDIs) when choosing a VMAT2 inhibitor for TD. AUSTEDO XR is primarily metabolized by CYP2D6, with minor contributions from CYP3A4/5 and other enzymes to form several minor metabolites.4 The metabolic profile of AUSTEDO XR allows for few restrictions related to DDIs.4 In patients taking concomitant strong CYP2D6 inhibitors or who are poor CYP2D6 metabolizers, there are 5 available dosing options with a maximum dosage of 36 mg/day.4 Only AUSTEDO XR allows for the ability to increase once-daily dose in patients taking strong CYP inhibitors and inducers.4,15
Moreover, no dose adjustments to P-glycoprotein (P-gp) substrates (eg, calcium channel blockers, statins, antimicrobials, and digoxin) are required when taking AUSTEDO XR (Figure 5).4,16 It is important to highlight that there are no specified restrictions on the use of AUSTEDO XR in patients taking concomitant anticholinergic medications.4
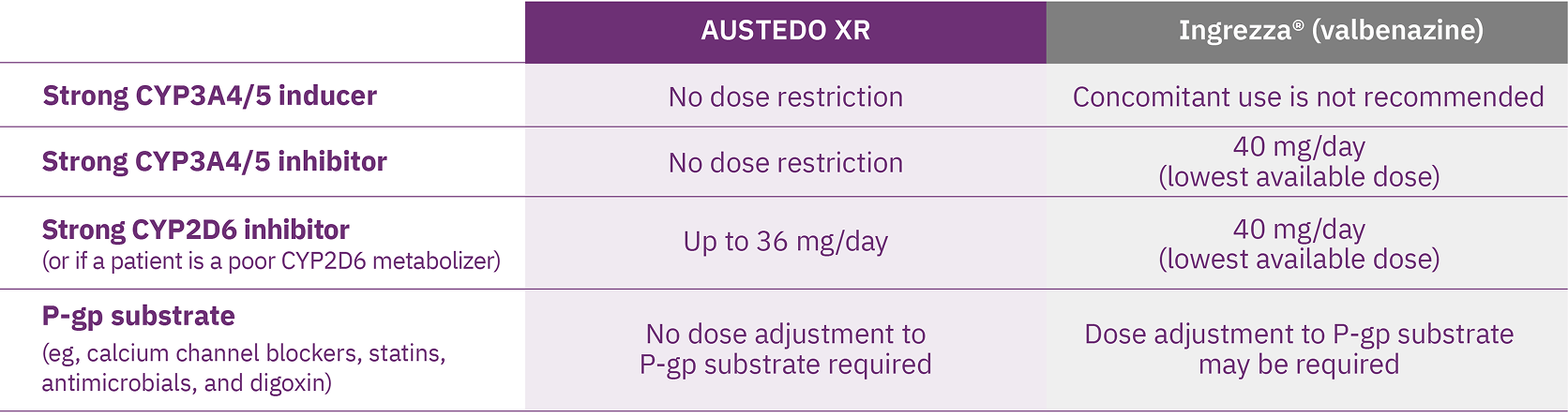
These differences should not be construed to imply differences in safety, efficacy, or clinical outcome.
Alongside clinical data, real-world evidence derived from patients' self-reported experiences can provide valuable insights to support discussions about treatment options with patients. A prospective, cross-sectional survey involving 209 patients with TD, which assessed patients' experiences, found that more than 90% of patients reported movement reduction with AUSTEDO XR, resulting in improvements in daily living.17 Patients also noted improved emotional, social, and physical well-being (Figure 6).17
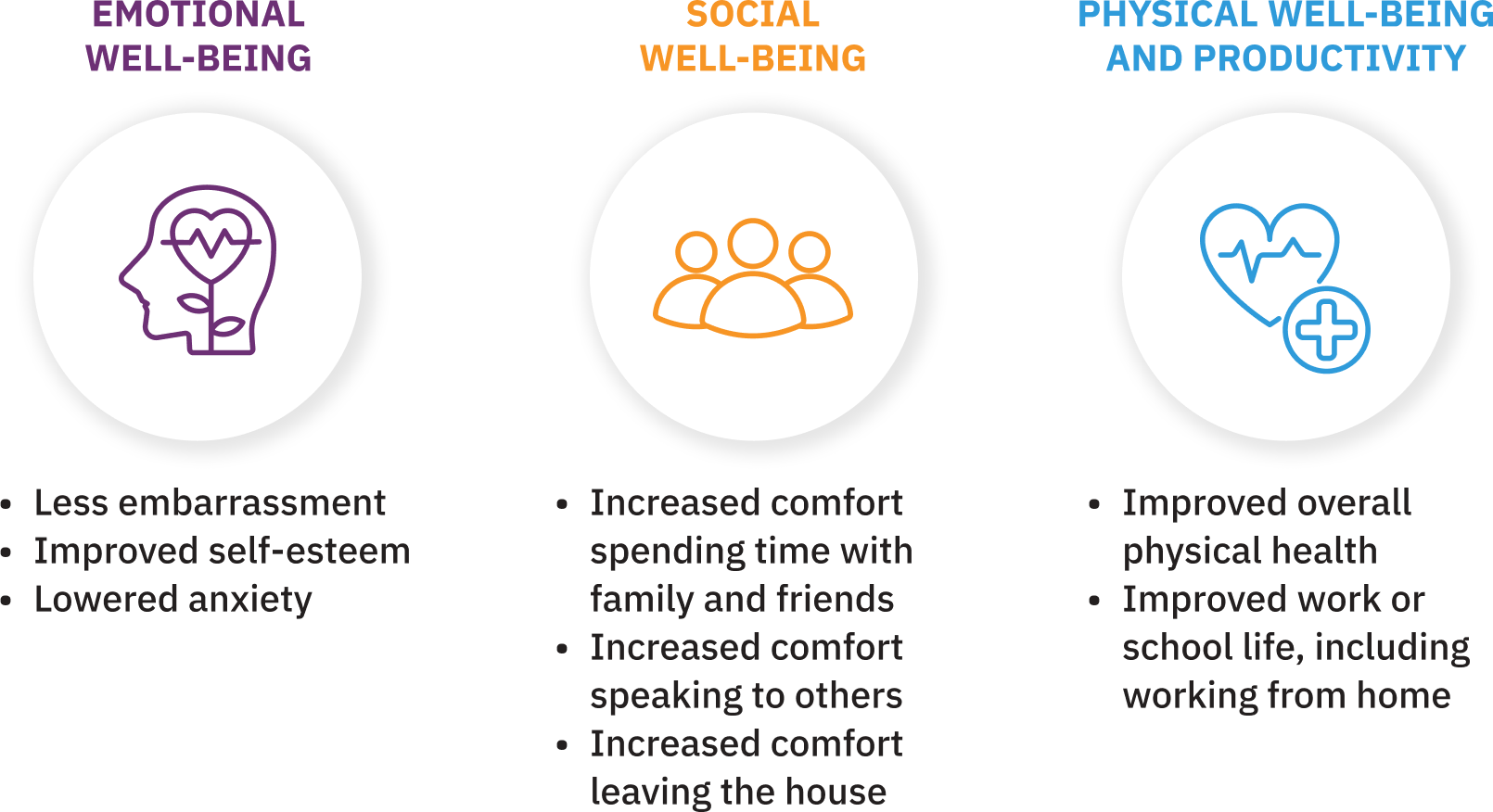
97% of patients planned to continue taking AUSTEDO XR. In addition, 60% reported taking it in the morning, and 42% reported usually taking it without food.17
- TD and DIP have distinct underlying pathophysiology and specific clinical presentation that require different treatment approaches1,2
- AUSTEDO XR is a VMAT2 inhibitor approved in the US for the treatment of adults with TD4
- Established efficacy and safety profile with AUSTEDO
- In placebo-controlled clinical trials, patients who received AUSTEDO BID demonstrated a rapid response as early as Week 2 and achieved a significant and meaningful reduction in TD severity at Week 124-6
- AUSTEDO demonstrated a significant improvement in AIMS reduction with a proven safety and tolerability profile across a diverse population over 3 years9-12
- Adverse reactions with AUSTEDO XR are expected to be similar to AUSTEDO4
- The flexible, once-daily AUSTEDO XR options enable patients to titrate as needed to achieve effective dosing and tolerable symptom control4
- Only AUSTEDO XR allows for the ability to increase once-daily dose in patients taking strong CYP inhibitors and inducers4,15
- Results from real-world surveys of patients with TD found improvements in daily living across several areas of their lives as a result of movement reduction with AUSTEDO XR17
1. Ward KM, Citrome L. Antipsychotic-related movement disorders: drug-induced parkinsonism vs. tardive dyskinesia—key differences in pathophysiology and clinical management. Neurol Ther. 2018;7(2):233-248. 2. Hauser RA, Meyer JM, Factor SA, et al. Differentiating tardive dyskinesia: a video-based review of antipsychotic-induced movement disorders in clinical practice. CNS Spectr. 2022;27(2):208-217. 3. American Psychiatric Association. The American Psychiatric Association Practice Guideline for the Treatment of Patients With Schizophrenia. 3rd ed. Washington, DC: American Psychiatric Association; 2021. Accessed November 4, 2025. https://psychiatryonline.org/doi/pdf/10.1176/appi.books.9780890424841 4. AUSTEDO XR® (deutetrabenazine) extended-release tablets/AUSTEDO® current Prescribing Information. Parsippany, NJ: Teva Neuroscience, Inc. 5. Anderson KE, Stamler D, Davis MD, et al. Deutetrabenazine for treatment of involuntary movements in patients with tardive dyskinesia (AIM-TD): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Psychiatry. 2017;4(8):595-604. 6. Fernandez HH, Factor SA, Hauser RA, et al. Randomized controlled trial of deutetrabenazine for tardive dyskinesia: the ARM-TD study. Neurology. 2017;88(21):2003-2010. 7. Data on file. Teva Neuroscience, Inc. Parsippany, NJ. 8. Benztropine mesylate tablet. Prescribing Information. Warren, NJ: Cipla USA, Inc. 9. Hauser RA, Barkay H, Fernandez HH, et al. Long-term deutetrabenazine treatment for tardive dyskinesia is associated with sustained benefits and safety: a 3-year, open-label extension study. Front Neurol. 2022;13:773999. 10. Nasrallah H, Chen M, Barkay H, Gordon MF, Finkbeiner S. Long-term efficacy and safety of deutetrabenazine in postmenopausal women with tardive dyskinesia. Presented at: 35th Annual Psych Congress 2022; September 17-20, 2022; New Orleans, LA. 11. Sajatovic M, Gandhi P, Konings M, Barash S, Finkbeiner S. Long-term safety and efficacy of deutetrabenazine in patients aged ≥65 years with tardive dyskinesia. Presented at: American Association for Geriatric Psychiatry; March 14-17, 2025; Phoenix, AZ. 12. Hauser RA, Barkay H, Fernandez HH, et al. Deutetrabenazine provides long-term benefit for tardive dyskinesia regardless of underlying condition and dopamine receptor antagonist use: a post hoc analysis of the 3-year, open-label extension study. J Clin Psychopharmacol. 2024;44(4):386-396. 13. Levi M, Schneider F, Gosselin NH, et al. Population pharmacokinetic and exposure safety analyses of deutetrabenazine in patients with moderate to severe tardive dyskinesia. Presented at: American Conference on Pharmacometrics; October 20-23, 2019; Orlando, FL. 14. Singh R, Sunzel EM, Dongwoo K, et al. Assessment of the deutetrabenazine exposure-response relationships for patients with moderate-to-severe tardive dyskinesia. Presented at: Psych Congress; September 17-20, 2022; New Orleans, LA. 15. Ingrezza® (valbenazine) capsules. Prescribing Information. San Diego, CA: Neurocrine Biosciences, Inc. 16. DrugBank Online. P-glycoprotein substrates. Accessed November 4, 2025. https://go.drugbank.com/categories/DBCAT002668 17. Jain R, Konings M, Thompson S, et al. Real-world evidence of patient experience with once-daily deutetrabenazine extended-release tablets for the treatment of tardive dyskinesia in the United States. Presented at: Annual Psych Congress Elevate; May 28-31, 2025; Las Vegas, NV.











