TD and DIP are common movement disorders caused by exposure to dopamine receptor blocking agents such as antipsychotics, antiemetics, and prokinetics. However, the symptoms associated with each disorder require different treatment approaches, as they result from opposite pathophysiological mechanisms (Video).1,2
- DIP is a hypodopaminergic disorder: the acute blockade of dopamine receptors by typical and atypical antipsychotics leads to a reduction in postsynaptic dopamine signaling. Reduced signaling results in the hypokinetic movements associated with DIP1,3
- TD is a hyperdopaminergic disorder: it is thought that chronic blockade of dopamine receptors results in an increase of receptor function, which is partly theorized to result from receptor hypersensitivity or upregulation. This results in increased dopamine signaling, manifesting as the hyperkinetic movements that are characteristic of TD1,2
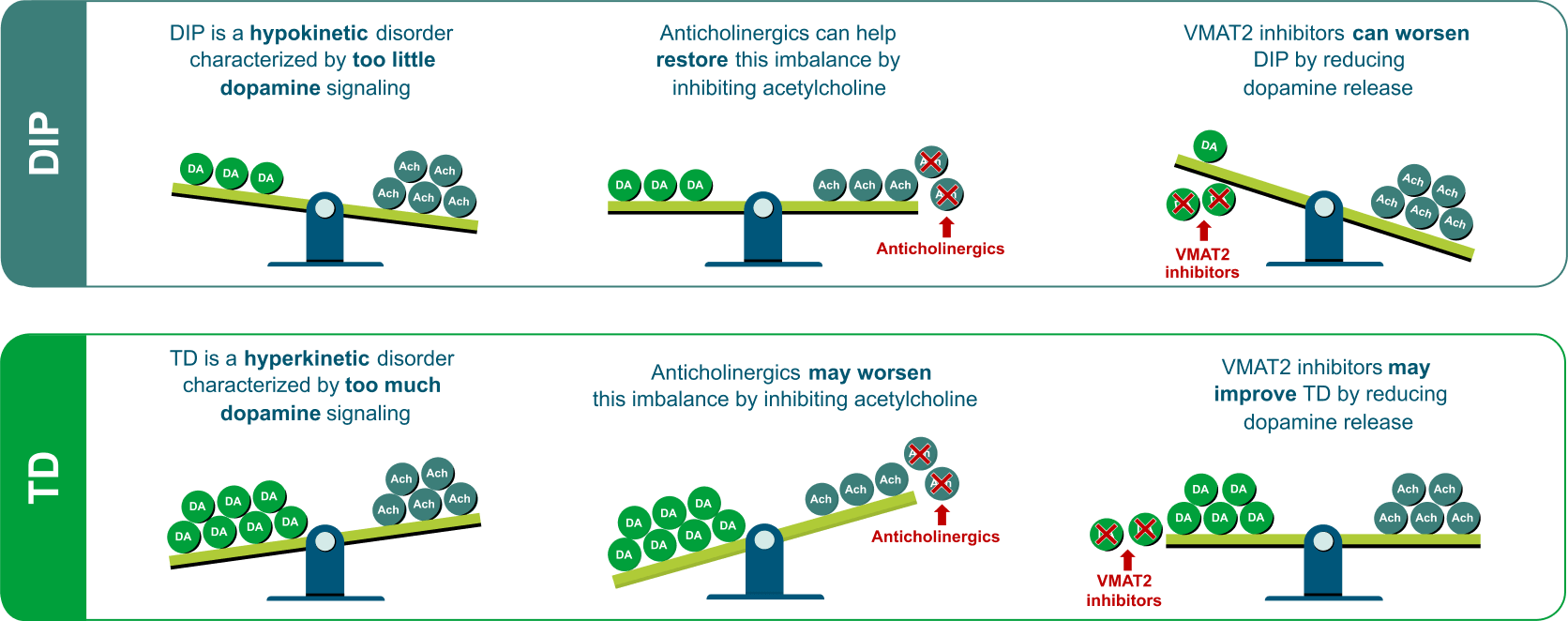
Differentiating between DIP and TD is crucial as the wrong treatment could lead to a worsening of symptoms.1,4 In DIP, a decrease in dopamine signaling amplifies cholinergic activity. Thus, anticholinergics can be effective in treating DIP because they inhibit acetylcholine and therefore restore the dopamine-acetylcholine balance. In contrast, in TD, an increase in dopamine signaling dampens cholinergic activity.5 Thus, anticholinergic medications such as benztropine are not effective in treating TD and may even worsen hyperkinetic symptoms because they increase acetylcholine signaling (Figure).1,4,6
Vesicular monoamine transporter 2 (VMAT2) inhibitors are the only US Food and Drug Administration (FDA)-approved treatment for TD and are recommended by the American Psychiatric Association (APA) Guidelines if TD has an impact on the patient, regardless of severity of movements.7 These agents have been shown to be effective in improving TD symptoms by reducing dopamine signaling and helping to restore the dopamine-acetylcholine balance.1 On the other hand, they can worsen DIP by further reducing dopamine signaling and amplifying cholinergic activity (Figure).1
For patients with TD who are taking an anticholinergic, gradually reduce and discontinue anticholinergic therapy and initiate treatment with a VMAT2 inhibitor as appropriate.1,5,7
- TD and DIP are different disorders with opposite pathophysiological mechanisms; DIP is a hypodopaminergic disorder while TD is a hyperdopaminergic disorder1-3
- Differentiating between DIP and TD is crucial as the wrong treatment could lead to a worsening of symptoms1,4
- Anticholinergic medications are indicated for the treatment of DIP but can exacerbate TD symptoms1,4
- VMAT2 inhibitors are indicated for TD but worsen parkinsonism1
- APA Guidelines recommend VMAT2 inhibitors for TD that has an impact on the patient, regardless of severity of movements7
1. Ward KM, Citrome L. Antipsychotic-related movement disorders: drug-induced parkinsonism vs. tardive dyskinesia—key differences in pathophysiology and clinical management. Neurol Ther. 2018;7(2):233-248. 2. Hauser RA, Meyer JM, Factor SA, et al. Differentiating tardive dyskinesia: a video-based review of antipsychotic-induced movement disorders in clinical practice. CNS Spectr. 2022;27(2):208-217. 3. Conn H, Jankovic J. Drug-induced parkinsonism: diagnosis and treatment. Expert Opin Drug Saf. 2024;23(12):1503-1513. 4. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed, Text Revision. Washington, DC: American Psychiatric Association; 2022. 5. Vanegas-Arroyave N, Caroff SN, Citrome L, et al. An evidence-based update on anticholinergic use for drug-induced movement disorders. CNS Drugs. 2024;38(4):239-254. 6. Benztropine mesylate tablet. Prescribing Information. Warren, NJ: Cipla USA Inc. 7. American Psychiatric Association. The American Psychiatric Association Practice Guideline for the Treatment of Patients With Schizophrenia. 3rd ed. Washington, DC: American Psychiatric Association; 2021.